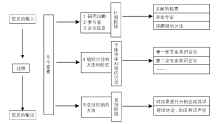
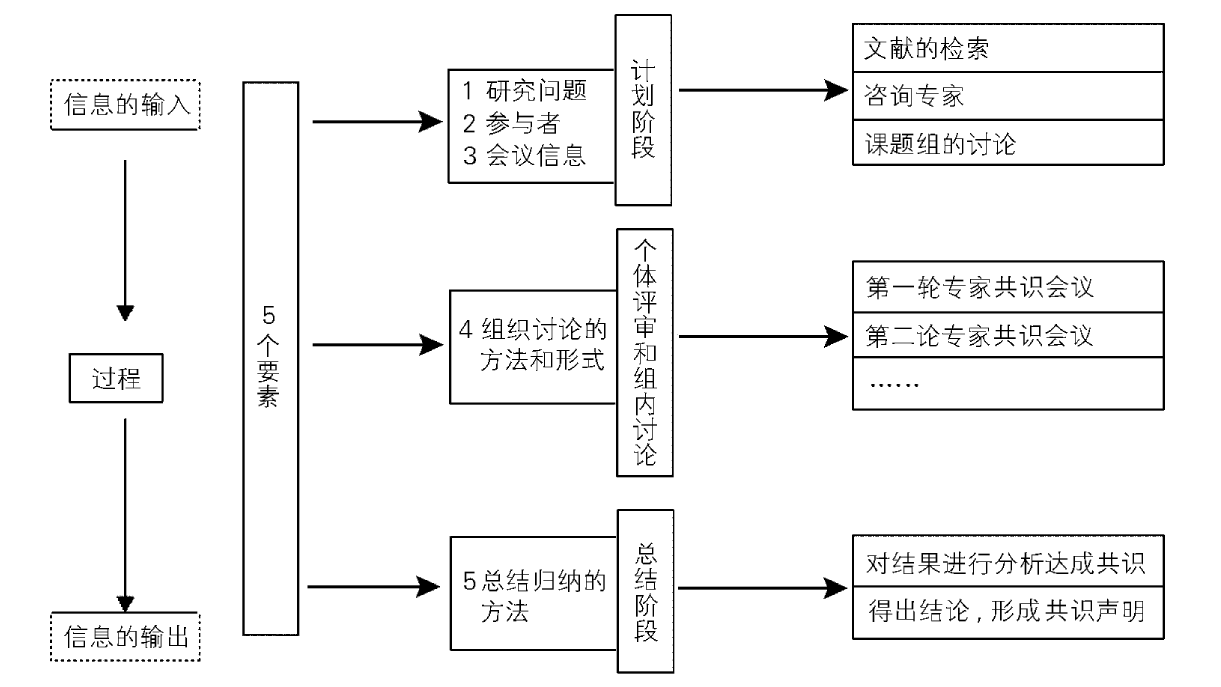
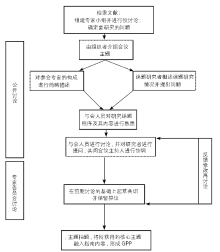
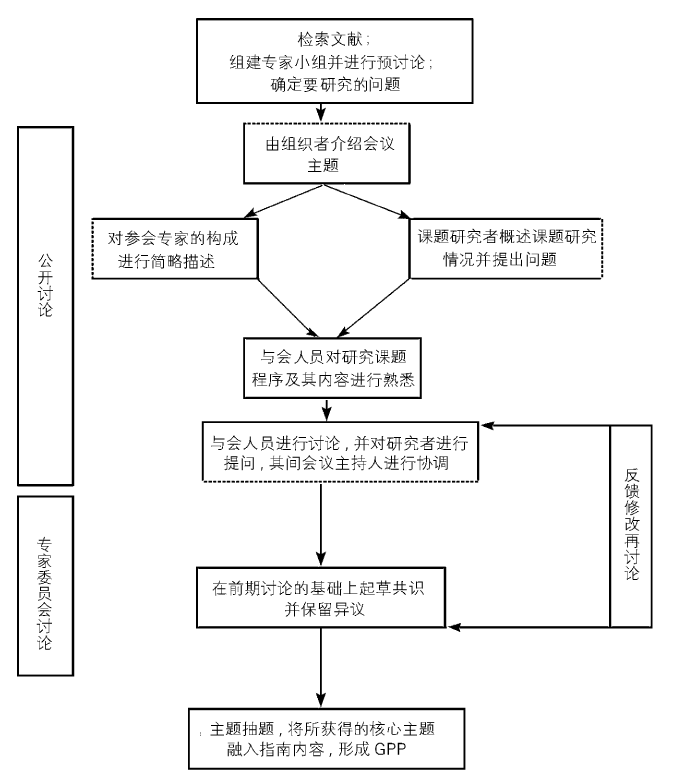
Journal of Chinese Integrative Medicine ›› 2008, Vol. 6 ›› Issue (6): 555-560.doi: 10.3736/jcim20080602
• Editorial • Previous Articles Next Articles
Application of consensus methods in making clinical practice guidelines of traditional medicine
- Institute of Basic Clinical Medicine, China Academy of Chinese Medical Sciences, Beijing 100700, China
CLC Number:
- R-3
| [1] | Moynihan R, Oxman AD, Lavis J , et al. A review of organizations that support the use of research evidence in developing guidelines, technology assessments, and health policy, for the WHO Advisory Committee on Health Research. Oslo, Norwegian Knowledge Centre for the Health Services. 2006 |
| [2] |
Burgers JS, Grol R, Klazinga NS , et al. Towards evidence-based clinical practice: an international survey of 18 clinical guideline programs[J]. Int J Qual Health Care, 2003,15(1):31-45
doi: 10.1093/intqhc/15.1.31 |
| [3] |
Murphy MK, Black NA, Lamping DL , et al. Consensus development methods and their use in clinical guideline development[J]. Health Technol Assess, 1998,2(3):1-88
doi: 10.4135/9781848608344 pmid: 9561895 |
| [4] |
Zhang BT . Evidence-based consensus statements and clinical guidelines: do the means meet the ends?[J]. Hongkong Med J, 2002,8(5):312
pmid: 12376705 |
| [5] | NIH consensus development program |
| [6] | NIH Consensus Development Program. Consensus development conferences: Guidelines for the planning and management of NIH consensus development conferences |
| [7] |
McGlynn EA, Kosecoff J, Brook RH . Format and conduct of consensus development conferences.Multi-nation comparison[J]. Int J Technol Assess Health Care, 1990,6(3):450-469
doi: 10.1017/S0266462300001045 |
| [8] |
Eccles M, Clapp Z, Grimshaw J , et al. North of England evidence based guidelines development project: methods of guideline development[J]. BMJ, 1996,312(7033):760-762
doi: 10.1136/bmj.312.7033.760 |
| [9] | Cochrane Methodology Register |
| [10] | National Guideline Clearinghouse . Agency for healthcare research and quality. 2007 |
| [11] | Guidelines International Network (GIN). Guidelines International Network |
| [12] |
Fretheim A, Schünemann HJ, Oxman AD . Improving the use of research evidence in guideline development: 5. Group processes[J]. Health Res Policy Syst, 2006,4:17
doi: 10.1186/1478-4505-4-17 |
| [13] |
Black NA, Murphy M, Lamping D , et al. Consensus development methods: a review of best practice in creating clinical guidelines[J]. J Health Serv Res Policy, 1999,4(4):236-248
doi: 10.1177/135581969900400410 |
| [14] | Pope C, Mays N. Qualitative research in health care(2nd ed)[M]. London, United Kingdom: BMJ Publishing Group,1999,(2nd ed): |
| [15] |
Jones J, Hunter D . Consensus methods for medical and health services research[J]. BMJ, 1995,311(7001):376-380
doi: 10.1136/bmj.311.7001.376 |
| [16] | Green J, Thorogood N . Qualitative methods for health research thousand[M]. London: Sage Publications, 2004 |
| [17] | Harbour R, Miller J . A new system for grading recommendations in evidence based guidelines[J]. BMJ, 2001,323(7308):334-336 |
| [18] |
Liu JP . The composition of evidence body of traditional medicine and recommendations for its evidence grading[J]. Zhongguo Zhong Xi Yi Jie He Za Zhi, 2007,27(12):1061-1062
doi: 10.3321/j.issn:1003-5370.2007.12.001 |
|
刘建平 . 传统医学证据体的构成及证据分级的建议[J]. 中国中西医结合杂志, 2007,27(12):1061-1062
doi: 10.3321/j.issn:1003-5370.2007.12.001 |
|
| [19] | Center for Evidence-Based Medicine. Levels of evidence |
| [20] | Scottish Intercollegiate Guidelines Network (SIGN). Systematic literature review |
| [21] |
Klibanski A, Adams-Campbell L, Bassford T , et al.NIH consensus development panel on osteoporosis prevention, diagnosis, therapy[J].JAMA, 2001, 285: 785-795
doi: 10.1001/jama.285.6.785 pmid: 11176917 |
| [22] |
Brown JP, Fortier M, Frame H . Canadian consensus conference on osteoporosis, 2006 update[J]. J Obstet Gynaecol Can, 2006,28(2 Suppl 1):S95-S112
doi: 10.1016/S1701-2163(16)32087-4 pmid: 16626523 |
| [23] | Australian National Consensus Conference 1996. The prevention and management of osteoporosis consensus statement. Med J Conference 1996. The prevention and management of osteoporosis consensus statement. Med[J] Aust. 1997; 167:S1-S15 |
| [24] |
Consensus development conference . prophylaxis and treatment of osteoporosis[J]. Am J Med, 1994,90(1):107-110
doi: 10.1007/BF01880454 pmid: 2739351 |
| [25] |
Peck WA . Consensus Development Conference: diagnosis, prophylaxis and treatment of osteoporosis[J]. Am J Med, 1993,94(6):646-650
doi: 10.1016/0002-9343(93)90218-E |
| [26] | Ministry of Health (Singapore).Osteoporosis[J].National Guideline Clearinghouse,2002,2:63 |
| Ministry of Health (Singapore).Osteoporosis[J].National Guideline Clearinghouse, 2002,2(63): | |
| [27] | Scottish Intercollegiate Guidelines Network (SIGN).Management of osteoporosis: a national clinical guideline[M].Edinburgh (Scotland): SIGN Publication, 2002: no62 |
| [28] | Western Pacific Region, World Health Organization . Clinical practice guideline of traditional medicine for primary osteoporosis. Zhongguo Zhong Yi Ke Xue Yuan. 2007 |
| 世界卫生组织西太区. 用于原发性骨质疏松症的传统医学临床实践指南讨论稿. 中国中医科学院. 2007 |
| [1] | Hui Luo, Jian-ping Liu, Qing Li. Methodological issues and suggestions for improvement in randomized controlled trials of Chinese herbal medicine for recurrent miscarriage. Journal of Chinese Integrative Medicine, 2012, 10(6): 604-614. |
| [2] | Xie Li-min, Wang Wen-yue. A brief introduction to Appraisal of Guidelines for Research and Evaluation Ⅱ. Journal of Chinese Integrative Medicine, 2012, 10(2): 160-165. |
| [3] | David Sibbritt, Jon Adams. Developing and promoting public health methods for integrative medicine: Examples from the field in Australia. Journal of Chinese Integrative Medicine, 2011, 9(3): 233-236. |
| [4] | Peng-fei Gao, Kenji Watanabe. Introduction of the World Health Organization project of the International Classification of Traditional Medicine. Journal of Chinese Integrative Medicine, 2011, 9(11): 1161-1164. |
| [5] | Hugh MacPherson , Douglas G Altman , Richard Hammerschlag , Youping Li , Taixiang Wu , Adrian White, David Moher . Revised STandards for Reporting Interventions in Clinical Trials of Acupuncture (STRICTA): Extending the CONSORT statement (Chinese version). Journal of Chinese Integrative Medicine, 2010, 8(9): 804-818. |
| [6] | Jian-xin Chen, Hui-hui Zhao, Wei Wang. Investigative strategy for research on biological basis of traditional Chinese medicine syndrome: feature selection-based data mining methods. Journal of Chinese Integrative Medicine, 2010, 8(8): 747-749. |
| [7] | David Moher, Sally Hopewell, Kenneth F Schulz, Victor Montori, Peter C Gtzsche, P J Devereaux, Diana Elbourne, Matthias Egger, Douglas G Altman. CONSORT 2010 Explanation and Elaboration: Updated guidelines for reporting parallel group randomised trials (Chinese version). Journal of Chinese Integrative Medicine, 2010, 8(8): 701-741. |
| [8] | Kenneth F Schulz, Douglas G Altman, David Moher, the CONSORT Group. CONSORT 2010 Statement: updated guidelines for reporting parallel group randomised trials (Chinese version). Journal of Chinese Integrative Medicine, 2010, 8(7): 604-612. |
| [9] | Xing Liao, Yan-ming Xie. Application of semi-structured and in-depth interview on optimization of clinical research program in traditional Chinese medicine. Journal of Chinese Integrative Medicine, 2009, 7(4): 309-314. |
| [10] | Ke-ji Chen, Yue-rong Jiang. Current status and problems in developing clinical guidelines for Chinese medicine and integrative medicine. Journal of Chinese Integrative Medicine, 2009, 7(4): 301-305. |
| [11] | Yan-ming Xie, Xing Liao. Quality control and assessment of qualitative interview in health care research. Journal of Chinese Integrative Medicine, 2008, 6(7): 668-673. |
| [12] | Jun-ying Fu. Current status of research on complementary and alternative medicine in the United States: in comparison with that on traditional Chinese medicine in China. Journal of Chinese Integrative Medicine, 2008, 6(6): 551-554. |
| [13] | Wei-xiong Liang. Problems-solving strategies in clinical treatment guideline for traditional Chinese medicine and integrative medicine. Journal of Chinese Integrative Medicine, 2008, 6(1): 1-4. |
| [14] | Jin-zhou Tian, Jing Shi, Xin-qing Zhang, Qi Bi, Xin Ma, Zhi-liang Wang, Xiao-bin Li, Shu-li Shen, Lin Li, Zhen-yun Wu, Li-yan Fang, Xiao-dong Zhao, Ying-chun Miao, Peng-wen Wang, Ying Ren, Jun-xiang Yin, Yong-yan Wang, Beijing United Study Group on MCI of the Capital Foundation of Medical Developments. An explanation on "guiding principles of clinical research on mild cognitive impairment (protocol)". Journal of Chinese Integrative Medicine, 2008, 6(1): 15-21. |
| [15] | Zhi-chun Jin. Problems in establishing clinical guideline for integrated traditional Chinese and Western medicine. Journal of Chinese Integrative Medicine, 2008, 6(1): 5-8. |