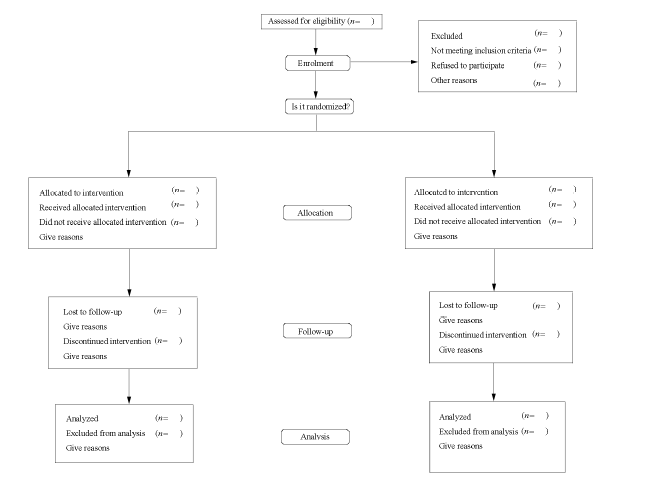
Journal of Chinese Integrative Medicine ›› 2006, Vol. 4 ›› Issue (5): 455-466.doi: 10.3736/jcim20060504
• Review • Previous Articles Next Articles
Critical appraisal of clinical studies in Chinese herbal medicine
Simon Dagenais1,2,3,Andrea C. Tricco1,4, Zhao-xiang Bian5,Wen-hua Huang6,David Moher1,2,3
- 1. Chalmers Research Group, Children's Hospital of Eastern Ontario Research Institute, Ottawa, Canada
2. Department of Pediatrics, Faculty of Medicine, University of Ottawa, Ottawa, Canada
3. Department of Epidemiology and Community Medicine, Faculty of Medicine, University of Ottawa, Ottawa, Canada
4. Institute of Population Health, University of Ottawa, Ottawa, Canada
5. School of Chinese Medicine, Hong Kong Baptist University, Hong Kong SAR, China
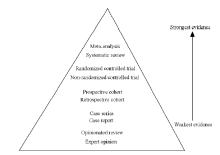
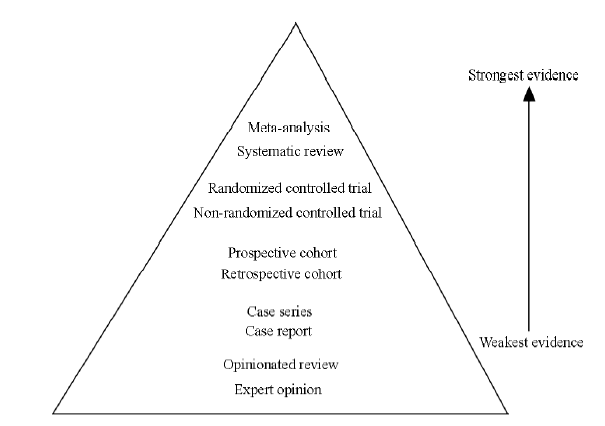
CLC Number:
- R-3
| [1] | Kessler RC, Davis RB, Foster DF , et al. Long-term trends in the use of complementary and alternative medical therapies in the United States[J]. Ann Intern Med, 2001,135(4):262-268 |
| [2] |
Tindle HA, Davis RB, Phillips RS , et al. Trends in use of complementary and alternative medicine by US adults: 1997-2002[J]. Altern Ther Health Med, 2005,11(1):42-49
doi: 10.1016/j.jep.2004.10.005 pmid: 15712765 |
| [3] | Schulz KF, Chalmers I, Hayes RJ , et al. Empirical evidence of bias. Dimensions of methodological quality associated with estimates of treatment effects in controlled trials[J]. JAMA, 1995,273(5):408-412 |
| [4] | Egger M, Juni P, Bartlett C , et al. How important are comprehensive literature searches and the assessment of trial quality in systematic reviews? Empirical study[J]. Health Technol Assess, 2003,7(1):1-6 |
| [5] | Sackett DL, Rosenberg WM, Gray JA , et al. Evidence based medicine: what it is and what it isn't[J]. BMJ, 1996,312(7023):71-72 |
| [6] | 6Meinert CL . Clinical trials: Design, conduct, and analysis(1st ed)[M]. New York: Oxford University Press,1986,(1st ed): |
| [7] | Bian ZX, Li YP, Moher D , et al. Improving the quality of randomized controlled trials in chinese herbal medicine, part I: clinical trial design and methodology[J]. Zhong Xi Yi Jie He Xue Bao, 2006,4(2):120-129 |
| [8] | Bian ZX, Moher D, Dagenais S , et al. Improving the quality of randomized controlled trials in Chinese herbal medicine, part II: control group design[J]. Zhong Xi Yi Jie He Xue Bao, 2006,4(2):130-136 |
| [9] | Evans D . Hierarchy of evidence: a framework for ranking evidence evaluating healthcare interventions[J]. J Clin Nurs, 2003,12(1):77-84 |
| [10] | Hulley S, Cumming SR, Browner WS, et al.Designing clinical research( 2nd edt ) [M].Philadelphia: Lippincott Williams & Wilkins, 2001,( 2nd edt): |
| [11] |
Mulrow CD . The medical review article: state of the science[J]. Ann Intern Med, 1987,106(13):485-488
doi: 10.7326/0003-4819-106-3-485 pmid: 3813259 |
| [12] | Mulrow CD . Rationale for systematic reviews[J]. BMJ, 1994,309(6954):597-599 |
| [13] | Cook DJ, Mulrow CD, Haynes RB . Systematic reviews: synjournal of best evidence for clinical decisions[J]. Ann Intern Med, 1997,126(5):376-380 |
| [14] | AGREE Collaboration . Development and validation of an international appraisal instrument for assessing the quality of clinical practice guidelines: the AGREE project[J]. Qual Safety Health Care, 2003,12(1):18-23 |
| [15] |
Gluud LL . Bias in clinical intervention research[J]. Am J Epidemiol, 2006,163(6):493-501
doi: 10.1093/aje/kwj069 pmid: 16443796 |
| [16] |
Tsay MY, Yang YH . Bibliometric analysis of the literature of randomized controlled trials[J]. J Med Libr Assoc, 2005,93(4):450-458
pmid: 16239941 |
| [17] | .[ OTHER].Plint A, Moher D, Altman DG, et al. Does the CONSORT checklist improve the quality of reports of randomized controlled trials? A systematic review. Presented at the 5th International Congress on Peer Review and Biomedical Communication ( Chicago, September 2005). |
| [18] | Begg C, Cho M, Eastwood S , et al. Improving the quality of reporting of randomized controlled trials. The CONSORT statement[J]. JAMA, 1996,276(8):637-639 |
| [19] | 19Plint AC, Moher D, Morrison A , et al. Does the CONSORT checklist improve the quality of reports of randomized controlled trials: a systematic review[J]. Med J Aust, 2006,185(5):263-267 |
| [20] | Moher D, Schulz KF , Altman DG.The CONSORT statement: revised recommendations for improving the quality of reports of parallel group randomized trials[J].BMC Med Res Methodol, 2001, 1():2. Epub 2001 Apr 20 |
| [21] | Freemantle N, Mason JM, Haines A , et al. CONSORT: an important step towards evidence-based health care. Consolidated Standards of Reporting Trials[J]. Ann Intern Med, 1997,126(1):81-83 |
| [22] | Schulz KF . The quest for unbiased research: randomized clinical trials and the CONSORT reporting guidelines[J]. Ann Neurol, 1997,41(5):569-573 |
| [23] |
Huston P, Hoey J . CMAJ endorses the CONSORT statement. CONsolidation of Standards for Reporting Trials[J]. CMAJ, 1996,155(9):1277-1282
doi: 10.1001/jama.276.8.649 pmid: 8911294 |
| [24] | Davidoff F . News from the International Committee of Medical Journal Editors[J]. Ann Intern Med, 2000,133(3):229-231 |
| [25] | Campbell MK, Elbourne DR, Altman DG , et al. CONSORT statement: extension to cluster randomised trials[J]. BMJ, 2004,328(7441):702-708 |
| [26] | Piaggio G, Elbourne DR, Altman DG , et al. Reporting of noninferiority and equivalence randomized trials: an extension of the CONSORT statement[J]. JAMA, 2006,295(10):1152-1160 |
| [27] |
Ioannidis JP, Evans SJ, Gotzsche PC , et al. Better reporting of harms in randomized trials: an extension of the CONSORT statement[J]. Ann Intern Med, 2004,141(10):781-788
doi: 10.7326/0003-4819-141-10-200411160-00009 pmid: 15545678 |
| [28] | Gagnier JJ, Boon H, Rochon P , et al. Reporting randomized, controlled trials of herbal interventions: an elaborated CONSORT statement[J]. Ann Intern Med, 2006,144(5):364-367 |
| [29] |
Moher D, Jadad AR, Tugwell P . Assessing the quality of randomized controlled trials. Current issues and future directions[J]. Int J Technol Assess Health Care, 1996,12(2):195-208
doi: 10.1017/S0266462300009570 pmid: 8707495 |
| [30] | Moher D, Jadad AR, Nichol G , et al. Assessing the quality of randomized controlled trials: an annotated bibliography of scales and checklists[J]. Control Clin Trials, 1995,16(1):62-73 |
| [31] | Verhagen AP, de Vet HC, de Bie RA , et al. The art of quality assessment of RCTs included in systematic reviews[J]. J Clin Epidemiol, 2001,54(7):651-654 |
| [32] | Jadad AR, Moore RA, Carroll D , et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary?[J]. Control Clin Trials, 1996,17(1):1-12 |
| [33] | Pildal J, Chan AW, Hrobjartsson A , et al.Comparison of descriptions of allocation concealment in trial protocols and the published reports: cohort study[J].BMJ, 2005, 330(7499):1049. Epub 2005 Apr 7 |
| Pildal J, Chan AW, Hrobjartsson A , et al.Comparison of descriptions of allocation concealment in trial protocols and the published reports: cohort study[J].BMJ, 2005, 330(7499):1049. Epub 2005 Apr 7. | |
| [34] |
Juni P, Witschi A, Bloch R , et al. The hazards of scoring the quality of clinical trials for meta-analysis[J]. JAMA, 1999,282(11):1054-1060
doi: 10.1001/jama.282.11.1054 pmid: 10493204 |
| [35] | Emerson JD, Burdick E, Hoaglin DC , et al. An empirical study of the possible relation of treatment differences to quality scores in controlled randomized clinical trials[J]. Control Clin Trials, 1990,11(5):339-352 |
| [36] | Moher D, Pham B, Jones A , et al. Does quality of reports of randomised trials affect estimates of intervention efficacy reported in meta-analyses?[J]. Lancet, 1998,352(9128):609-613 |
| [37] | .[ OTHER]. 37Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions 4.2.5. 2005. THER].37Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions 4.2.5. 2005. .37Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions 4.2.5. 2005. THER].37Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions 4.2.5. 2005. |
| [38] | Leung KSY, Bian ZX, Moher D , et al. Improving the quality of randomized controlled trials in Chinese herbal medicine, part Ⅲ: quality control of Chinese herbal medicine used in randomized controlled trials[J]. Zhong Xi Yi Jie He Xue Bao, 2006,4(3):225-232 |
| [1] | Jing-lin Shi, Wan-hong Miao. Effects of acupuncture on lactoferrin content in tears and tear secretion in patients suffering from dry eyes: A randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(9): 1003-1008. |
| [2] | Jian-sheng Li, Ya Li, Su-yun Li, Yuan-yuan Wang, Li Deng, Yan-ge Tian, Su-li Jiang , Ying Wang. Long-term effects of Tiaobu Feishen therapies on systemic and local inflammation responses in rats with stable chronic obstructive pulmonary disease. Journal of Chinese Integrative Medicine, 2012, 10(9): 1039-1048. |
| [3] | Ming T. Tan , Jian-ping Liu , Lixing Lao. Adequate application of quantitative and qualitative statistic analytic methods in acupuncture clinical trials. Journal of Chinese Integrative Medicine, 2012, 10(8): 847-852. |
| [4] | Lu Yu, Shui-miao Lin, Ru-qian Zhou, Wei-jun Tang, Pin-xian Huang, Ying Dong, Jian Wang, Zhi-hua Yu, Jiu-lin Chen, Li Wei, San-li Xing, Hui-juan Cao, Hong-bin Zhao. Chinese herbal medicine for patients with mild to moderate Alzheimer disease based on syndrome differentiation: A randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(7): 766-776. |
| [5] | Ling Xu, Qiao-ling Zhou, Jie Han, Dan-ying Zhang, Hui-juan Dong, Zai-long Cai, Chao-qin Yu, Dong-xia Zhai . Effects of Chinese herbal medicine Neiyi Recipe-medicated serum on angiopoiesis of endometriosis in the chick chorioallantoic membrane model. Journal of Chinese Integrative Medicine, 2012, 10(7): 800-806. |
| [6] | Dong-mei Zhang, Li-xia Lou, Ai-ming Wu, Xi-ying Lü, Zhao-juan Hu, Yan-hong Zhang, Hong-fang Liu. Effects of Astragalus membranaceus and Potentilla discolor mixture on insulin resistance and its related mRNA expressions in KKAy mice with type 2 diabetes. Journal of Chinese Integrative Medicine, 2012, 10(7): 821-826. |
| [7] | Mei-jiao Mao, Jun-ping Hu, Cong Wang, Yi-yi Zhang, Ping Liu. Effects of Chinese herbal medicine Guanxinkang on expression of PPARγ-LXRα-ABCA1 pathway in ApoE-knockout mice with atherosclerosis. Journal of Chinese Integrative Medicine, 2012, 10(7): 814-820. |
| [8] | Yi-tao Zhao , Xin-guang Zhang , Li Bai , Li-qing Li , Jian-er Yu . Effects of Chinese herbal medicine Pingchuan Formula on airway inflammation, interferon-γ and interleukin-4 in mice with asthma. Journal of Chinese Integrative Medicine, 2012, 10(7): 807-813. |
| [9] | Ming-xing Zhao, Zhen-hua Dong, Zhong-hai Yu, Shi-yuan Xiao, Ya-ming Li . Effects of Ginkgo biloba extract in improving episodic memory of patients with mild cognitive impairment: A randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(6): 628-634. |
| [10] | Shui-qiu Deng, Xue-nong Ouyang, Zong-yang Yu, Xi-hu Dai, Xi Chen, Fang-zheng Fang, Wen-wu Wang, Zhi-zhen Liu. Influence of Chinese herbal medicine Feitai Capsule on completion or delay of chemotherapy in patients with stage ⅢB/Ⅳ non-small-cell lung cancer: a Randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(6): 635-640. |
| [11] | Chuan Chen, Hui-ying Chi, Zhi-hua Yu, Jiu-lin Chen . Effects of Chinese herbal medicine Shoushen Granule on telomere length and telomerase activity of peripheral white blood cells and vascular cells in rats with atherosclerosis. Journal of Chinese Integrative Medicine, 2012, 10(6): 667-673. |
| [12] | Hui Luo, Jian-ping Liu, Qing Li. Methodological issues and suggestions for improvement in randomized controlled trials of Chinese herbal medicine for recurrent miscarriage. Journal of Chinese Integrative Medicine, 2012, 10(6): 604-614. |
| [13] | Peng Fu , Xue-qiang Huang, Ai-hong Yuan, Guang Yu, Xiao-bin Mei, Ruo-lan Cu. Effects of salvianolate combined with alprostadil and reduced glutathione on progression of chronic renal failure in patients with chronic kidney diseases: a long-term randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(6): 641-646. |
| [14] | Daniel A. Weber, Janelle M. Wheat , Geoffrey M. Currie. Cancer stem cells and the impact of Chinese herbs, isolates and other complementary medical botanicals: A review. Journal of Chinese Integrative Medicine, 2012, 10(5): 493-503. |
| [15] | Qing-lan Wang, Yan-yan Tao , Li Shen , Hong-yan Cui , Cheng-hai Liu. Chinese herbal medicine Fuzheng Huayu recipe inhibits liver fibrosis by mediating the transforming growth factor-β1/Smads signaling pathway. Journal of Chinese Integrative Medicine, 2012, 10(5): 561-568. |