Journal of Chinese Integrative Medicine ›› 2010, Vol. 8 ›› Issue (7): 604-612.doi: 10.3736/jcim20100702
• Biomedical Publication • Previous Articles Next Articles
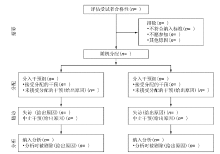
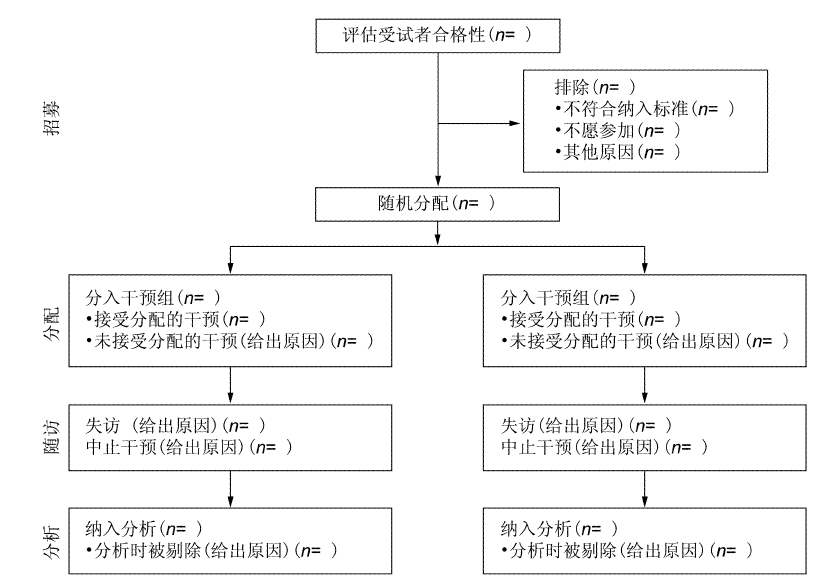
CONSORT 2010 Statement: updated guidelines for reporting parallel group randomised trials (Chinese version)
Kenneth F Schulza ,Douglas G Altmanb,David Moherc,the CONSORT Group
,Douglas G Altmanb,David Moherc,the CONSORT Group
- a Family Health International, Research Triangle Park, NC 27709, USA
b Centre for Statistics in Medicine, University of Oxford,Wolf son College,Oxford,UK
c Ottawa Methods Centre, Clinical Epidemiology Program, Ottawa Hospital Research Institute, Department of Epidemiology and Community Medicine, University of Ottawa, Ottawa, Canada
| [1] |
Jüni P, Altman DG, Egger M . Systematic reviews in health care: assessing the quality of controlled clinical trials[J]. BMJ, 2001,323(7303):42-46
doi: 10.1136/bmj.323.7303.42 pmid: 11440947 |
| [2] |
Chan AW, Altman DG . Epidemiology and reporting of randomised trials published in PubMed journals[J]. Lancet, 2005,365(9465):1159-1162
doi: 10.1016/S0140-6736(05)71879-1 |
| [3] |
Glasziou P, Meats E, Heneghan C, Shepperd S . What is missing from descriptions of treatment in trials and reviews?[J]. BMJ, 2008,336(7659):1472-1474
doi: 10.1136/bmj.39590.732037.47 pmid: 2440840 |
| [4] |
Dwan K, Altman DG, Arnaiz JA, Bloom J, Chan AW, Cronin E, Decullier E, Easterbrook PJ, Von Elm E, Gamble C, Ghersi D, Ioannidis JP, Simes J, Williamson PR . Systematic review of the empirical evidence of study publication bias and outcome reporting bias[J]. PLoS One, 2008,3(8):e3081
doi: 10.1371/journal.pone.0003081 |
| [5] |
Begg C, Cho M, Eastwood S, Horton R, Moher D, Olkin I, Pitkin R, Rennie D, Schulz KF, Simel D, Stroup DF . Improving the quality of reporting of randomized controlled trials. The CONSORT statement[J]. JAMA, 1996,276(8):637-639
doi: 10.1001/jama.1996.03540080059030 |
| [6] |
Moher D, Schulz KF, Altman DG . The CONSORT statement: revised recommendations for improving the quality of reports of parallel-group randomised trials[J]. Lancet, 2001,357(9263):1191-1194
doi: 10.1016/S0140-6736(00)04337-3 |
| [7] |
Moher D, Schulz KF, Altman DG . The CONSORT statement: revised recommendations for improving the quality of reports of parallel-group randomized trials[J]. Ann Intern Med, 2001,134(8):657-662
doi: 10.7326/0003-4819-134-8-200104170-00011 |
| [8] |
Moher D, Schulz KF, Altman D . The CONSORT statement: revised recommendations for improving the quality of reports of parallel-group randomized trials[J]. JAMA, 2001,285(15):1987-1991
doi: 10.1001/jama.285.15.1987 |
| [9] | Plint AC, Moher D, Morrison A, Schulz K, Altman DG, Hill C, Gaboury Ⅰ . Does the CONSORT checklist improve the quality of reports of randomised controlled trials? A systematic review[J]. Med J Aust, 2006,185(5):263-267 |
| [10] |
Hopewell S, Dutton S, Yu LM, Chan AW , Altman DG.The quality of reports of randomised trials in 2000 and 2006: comparative study of articles indexed by PubMed[J]. BMJ,2010,340: c723
doi: 10.1136/bmj.c723 |
| [11] |
Campbell MK, Elbourne DR, Altman DG; CONSORT group.CONSORT statement: extension to cluster randomised trials[J]. BMJ, 2004,328(7441):702-708
doi: 10.1136/bmj.328.7441.702 |
| [12] |
Piaggio G, Elbourne DR, Altman DG, Pocock SJ, Evans SJ . Reporting of noninferiority and equivalence randomized trials: an extension of the CONSORT statement[J]. JAMA, 2006,295(10):1152-1160
doi: 10.1001/jama.295.10.1152 |
| [13] |
Moher D, Hopewell S, Schulz KF, Montori V, Gtzsche PC, Devereaux PJ, Elbourne D, Egger M, Altman DG.CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials[J]. BMJ,2010,340: c869
doi: 10.1136/bmj.c869 |
| [14] |
Schulz KF, Chalmers I, Hayes RJ, Altman DG . Empirical evidence of bias. Dimensions of methodological quality associated with estimates of treatment effects in controlled trials[J]. JAMA, 1995,273(5):408-412
doi: 10.1001/jama.1995.03520290060030 |
| [15] |
Moher D, Pham B, Jones A, Cook DJ, Jadad AR, Moher M, Tugwell P, Klassen TP . Does quality of reports of randomised trials affect estimates of intervention efficacy reported in meta-analyses?[J]. Lancet, 1998,352(9128):609-613
doi: 10.1016/S0140-6736(98)01085-X |
| [16] |
Moher D, Liberati A, Tetzlaff J, Altman DG, for the PRISMA Group.Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement[J].BMJ , 2009, 339: b2535
doi: 10.14306/renhyd.18.3.114 pmid: 21145753 |
| [17] |
Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, Irwig LM, Lijmer JG, Moher D , Rennie D, de Vet HC, Standards for Reporting of Diagnostic Accuracy.Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD initiative[J]. BMJ, 2003,326(7379):41-44
doi: 10.1136/bmj.326.7379.41 |
| [18] |
von Elm E, Altman DG, Egger M, Pocock SJ, Gtzsche PC, Vandenbroucke JP, for the STROBE Initiative . Strengthening the reporting of observational studies in epidemiology(STROBE) statement: guidelines for reporting observational studies[J]. BMJ, 2007,335(7624):806-808
doi: 10.1136/bmj.39335.541782.AD |
| [19] |
Standards of Reporting Trials Group. A proposal for structured reporting of randomized controlled trials[J]. JAMA, 1994,272(24):1926-1931
doi: 10.1001/jama.1994.03520240054041 |
| [20] |
Rennie D . Reporting randomized controlled trials. An experiment and a call for responses from readers[J]. JAMA, 1995,273(13):1054-1055
doi: 10.1001/jama.1995.03520370096044 |
| [21] |
Hopewell S, Clarke M, Moher D, Wager E, Middleton P, Altman DG, Schulz KF, CONSORT Group . CONSORT for reporting randomised trials in journal and conference abstracts[J]. Lancet, 2008,371(9609):281-283
doi: 10.1016/S0140-6736(07)61835-2 pmid: 18221781 |
| [22] |
Chan AW , Hr bjartsson A, Haahr MT, G tzsche PC, Altman DG.Empirical evidence for selective reporting of outcomes in randomized trials: comparison of protocols to published articles[J]. JAMA, 2004,291(20):2457-2465
doi: 10.1001/jama.291.20.2457 |
| [23] |
Sackett DL . Commentary: Measuring the success of blinding in RCTs: don’t, must, can’t or needn’t?[J]. Int J Epidemiol, 2007,36(3):664-665
doi: 10.1093/ije/dym088 |
| [24] |
Schulz KF, Grimes DA . Blinding in randomised trials: hiding who got what[J]. Lancet, 2002,359(9307):696-700
doi: 10.1016/S0140-6736(02)07816-9 pmid: 11879884 |
| [25] |
Montori VM, Devereaux PJ, Adhikari NK, Burns KE, Eggert CH, Briel M, Lacchetti C, Leung TW, Darling E, Bryant DM, Bucher HC, Schünemann HJ, Meade MO, Cook DJ, Erwin PJ, Sood A, Sood R, Lo B, Thompson CA, Zhou Q, Mills E, Guyatt GH . Randomized trials stopped early for benefit: a systematic review[J]. JAMA, 2005,294(17):2203-2209
doi: 10.1001/jama.294.17.2203 |
| [26] |
Hollis S, Campbell F . What is meant by intention to treat analysis? Survey of published randomised controlled trials[J]. BMJ, 1999,319(7211):670-674
doi: 10.1136/bmj.319.7211.670 pmid: 10480822 |
| [27] |
Nuovo J, Melnikow J, Chang D . Reporting number needed to treat and absolute risk reduction in randomized controlled trials.[J]. JAMA, 2002,287(21):2813-2814
doi: 10.1001/jama.287.21.2813 pmid: 12038920 |
| [28] |
Ioannidis JP, Evans SJ , G tzsche PC, O’Neill RT, Altman DG, Schulz K, Moher D; CONSORT Group.Better reporting of harms in randomized trials: an extension of the CONSORT statement[J]. Ann Intern Med, 2004,141(10):781-788
doi: 10.7326/0003-4819-141-10-200411160-00009 pmid: 15545678 |
| [29] |
De Angelis C, Drazen JM, Frizelle FA, Haug C, Hoey J, Horton R, Kotzin S, Laine C, Marusic A, Overbeke AJ, Schroeder TV, Sox HC, Weyden MB; International Committee of Medical Journal Editors.Clinical trial registration: a statement from the International Committee of Medical Journal Editors[J]. Lancet, 2004,364(9438):911-912
doi: 10.1016/S0140-6736(04)17034-7 |
| [30] |
Lexchin J, Bero LA, Djulbegovic B, Clark O . Pharmaceutical industry sponsorship and research outcome and quality: systematic review[J]. BMJ, 2003,326(7400):1167-1170
doi: 10.1136/bmj.326.7400.1167 pmid: 156458 |
| [31] |
Hopewell S, Clarke M, Moher D, Wager E, Middleton P, Altman DG, Schulz KF, CONSORT Group . CONSORT for reporting randomized controlled trials in journal and conference abstracts: explanation and elaboration[J]. PLoS Med, 2008,5(1):e20
doi: 10.1371/journal.pmed.0050020 |
| [32] |
Boutron I, Moher D, Altman DG, Schulz KF, Ravaud P . Extending the CONSORT statement to randomized trials of nonpharmacologic treatment: explanation and elaboration[J]. Ann Intern Med, 2008,148(4):295-309
doi: 10.7326/0003-4819-148-4-200802190-00008 |
| [33] |
Gagnier JJ, Boon H, Rochon P, Moher D, Barnes J, Bombardier C . Reporting randomized, controlled trials of herbal interventions: an elaborated CONSORT statement[J]. Ann Intern Med, 2006,144(5):364-367
doi: 10.7326/0003-4819-144-5-200603070-00013 |
| [34] | Zwarenstein M, Treweek S, Gagnier JJ, Altman DG, Tunis S, Haynes B, Oxman AD, Moher D , CONSORT group, Pragmatic Trials in Healthcare (Practihc) group.Improving the reporting of pragmatic trials: an extension of the CONSORT statement[J]. BMJ, 2008,11(337):a2390 |
| [1] | Yoshiharu Motoo, Takashi Hakamatsuka, Nobuo Kawahara, Ichiro Arai, Kiichiro Tsutani. Standards of Reporting Kampo Products (STORK) in research articles. Journal of Integrative Medicine, 2017, 15(3): 182-185. |
| [2] | Zhang-ling Zhou, Cheng-xin Li, Yue-bo Jiang, Cong Zuo, Yun Cai, Rui Wang. Correlation between facial nerve functional evaluation and efficacy evaluation of acupuncture treatment for Bell’s palsy. Journal of Chinese Integrative Medicine, 2012, 10(9): 997-1002. |
| [3] | Lu Liu, Ying Gao. Study on the correlation between traditional Chinese medicine syndrome and short-term prognosis of ischemic stroke using logistic regression model and repeated-measures analysis of variance. Journal of Chinese Integrative Medicine, 2012, 10(9): 983-990. |
| [4] | Jing-lin Shi, Wan-hong Miao. Effects of acupuncture on lactoferrin content in tears and tear secretion in patients suffering from dry eyes: A randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(9): 1003-1008. |
| [5] | Oing-na Li , Xiu-ling Huang , Rui Gao , Fang Lu. Discussion on developing a data management plan and its key factors in clinical study based on electronic data capture system. Journal of Chinese Integrative Medicine, 2012, 10(8): 841-846. |
| [6] | Ming T. Tan , Jian-ping Liu , Lixing Lao. Adequate application of quantitative and qualitative statistic analytic methods in acupuncture clinical trials. Journal of Chinese Integrative Medicine, 2012, 10(8): 847-852. |
| [7] | Chen Xia , Qun-bang Zhu, Feng Huang, Jing-hui Huang, Hong-da Chen, Ming-jun Chen, Wei Gu, Bai Li, Fan-fu Fang, Liang-neng Wu . Traditional Chinese medicine constitution types in 127 elderly patients with insomnia: An investigation in communities of Yangpu District, Shanghai. Journal of Chinese Integrative Medicine, 2012, 10(8): 866-873. |
| [8] | Lu Yu, Shui-miao Lin, Ru-qian Zhou, Wei-jun Tang, Pin-xian Huang, Ying Dong, Jian Wang, Zhi-hua Yu, Jiu-lin Chen, Li Wei, San-li Xing, Hui-juan Cao, Hong-bin Zhao. Chinese herbal medicine for patients with mild to moderate Alzheimer disease based on syndrome differentiation: A randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(7): 766-776. |
| [9] | Hui Luo, Jian-ping Liu, Qing Li. Methodological issues and suggestions for improvement in randomized controlled trials of Chinese herbal medicine for recurrent miscarriage. Journal of Chinese Integrative Medicine, 2012, 10(6): 604-614. |
| [10] | Peng Fu , Xue-qiang Huang, Ai-hong Yuan, Guang Yu, Xiao-bin Mei, Ruo-lan Cu. Effects of salvianolate combined with alprostadil and reduced glutathione on progression of chronic renal failure in patients with chronic kidney diseases: a long-term randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(6): 641-646. |
| [11] | Shui-qiu Deng, Xue-nong Ouyang, Zong-yang Yu, Xi-hu Dai, Xi Chen, Fang-zheng Fang, Wen-wu Wang, Zhi-zhen Liu. Influence of Chinese herbal medicine Feitai Capsule on completion or delay of chemotherapy in patients with stage ⅢB/Ⅳ non-small-cell lung cancer: a Randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(6): 635-640. |
| [12] | Ming-xing Zhao, Zhen-hua Dong, Zhong-hai Yu, Shi-yuan Xiao, Ya-ming Li . Effects of Ginkgo biloba extract in improving episodic memory of patients with mild cognitive impairment: A randomized controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(6): 628-634. |
| [13] | Christoph A. Doenitz, Ana Anjos, Thomas Efferth , Tobias Greten, Henry J. Greten. Can heat and cold be parameterized? Clinical data of a preliminary study. Journal of Chinese Integrative Medicine, 2012, 10(5): 532-537. |
| [14] | Qin-hui Fu, Jian Pei, Qi Jia, Yi Song, Yue-hua Gu, Xiao-xin You . Acupuncture treatment programs for post-stroke motor rehabilitation in community hospitals: Study protocol of a multicenter, randomized, controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(5): 516-524. |
| [15] | Ying-chun Miao , Jin-zhou Tian , Jing Shi , Min Mao. Effects of Chinese medicine for tonifying the kidney and resolving phlegm and blood stasis in treating patients with amnestic mild cognitive impairment: A randomized, double-blind and parallel-controlled trial. Journal of Chinese Integrative Medicine, 2012, 10(4): 390-397. |
|