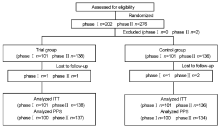
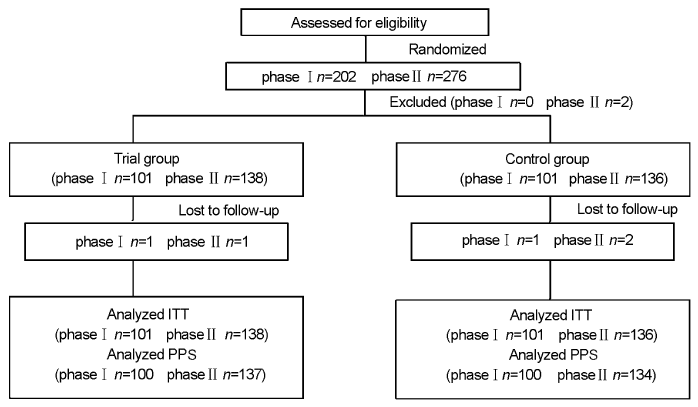
Journal of Chinese Integrative Medicine ›› 2008, Vol. 6 ›› Issue (12): 1238-1245.doi: 10.3736/jcim20081206
• Original Clinical Research • Previous Articles Next Articles
Andrographolide drop-pill in treatment of acute upper respiratory tract infection with external wind-heat syndrome: a multicenter and randomized controlled trial
Jing Chang1, Rui-ming Zhang1( ), Ying Zhang1, Zhi-bin Chen2, Zong-ming Zhang3, Qiang Xu4, Yu-ping Yang5, You-yu Long6, Liang-li Liu7, Hong-yan Cai8, Jie Cai9, Nan Lu10, Bing Mao1, Lei Wang1, Ting-qian Li1
), Ying Zhang1, Zhi-bin Chen2, Zong-ming Zhang3, Qiang Xu4, Yu-ping Yang5, You-yu Long6, Liang-li Liu7, Hong-yan Cai8, Jie Cai9, Nan Lu10, Bing Mao1, Lei Wang1, Ting-qian Li1
- 1. Department of Integrated Traditional Chinese and Western Medicine, West China Hospital, Sichuan University, Chengdu, Sichuan Province 610041, China
2. Department of Respiratory Diseases, Fujian Academy of Traditional Chinese Medicine, Fuzhou,Fujian Province 350108, China
3. Department of Respiratory Diseases, the First Affiliated Hospital, Anhui College of Traditional Chinese Medicine, Hefei, Anhui Province 230031, China
4. Department of Respiratory Diseases, Affiliated Hospital, Anhui Medical University, Hefei, Anhui Province 230022, China
5. Department of Respiratory Diseases, Affiliated Hospital, Jiangxi College of Traditional Chinese Medicine, Nanchang, Jiangxi Province 330006, China
6. Department of Respiratory Diseases, the Second Affiliated Hospital, Tianjin College of Traditional Chinese Medicine, Tianjin 300150, China
7. Department of Respiratory Diseases, the First Affiliated Hospital, Guiyang College of Traditional Chinese Medicine, Guiyang,Guizhou Province 550001,China
8. Department of Respiratory Diseases, Jilin Academy of Traditional Chinese Medicine, Changchun, Jilin Province 130021, China
9. Department of Respiratory Diseases, Affiliated Hospital, Shaanxi College of Traditional Chinese Medicine, Xi'an, Shaanxi Province 712083,China
10. Department of Respiratory Diseases, General Hospital of Beijing Military Region, Beijing 100700, China
CLC Number:
- R511.6
| [1] |
Li SG, Ye ZY . Pharmacological activity of andrographolide[J]. Zhonghua Zhong Yi Yao Xue Kan, 2008,26(5):984-986
doi: 10.3969/j.issn.1673-7717.2008.05.034 |
|
李曙光, 叶再元 . 穿心莲内酯的药理活性作用[J]. 中华中医药学刊, 2008,26(5):984-986
doi: 10.3969/j.issn.1673-7717.2008.05.034 |
|
| [2] |
Chen JH, Fu WM . Clinical observation of andrograp-holide tablet in treating upper respiratory tract infection[J]. Zhongguo Zhong Yi Ji Zheng, 2004,13(2):87
doi: 10.3969/j.issn.1004-745X.2004.02.012 |
|
陈炯华, 符为民 . 穿心莲内酯片治疗上呼吸道感染临床观察[J]. 中国中医急症, 2004,13(2):87
doi: 10.3969/j.issn.1004-745X.2004.02.012 |
|
| [3] | Wang JY . Comparison experiment of the release of andrographolide drop-pill and andrographolide tablet[J]. Henan Zhong Yi Yao Xue Kan, 2001,17(4):29 |
| 王景云 . 穿心莲滴丸及穿心莲内酯片释放度的比较实验[J]. 河南中医药学刊, 2001,17(4):29 | |
| [4] |
Chang J, Li TQ, Wan MH , et al. Quality assessment for randomized controlled trials published in four acta of traditional Chinese medicine[J]. Zhongguo Xun Zheng Yi Xue Za Zhi, 2006,6(3):171-178
doi: 10.3969/j.issn.1672-2531.2006.03.004 |
|
常静, 李廷谦, 万美华 , 等. 中国4种中医药大学学报随机对照临床试验文献的质量评价[J]. 中国循证医学杂志, 2006,6(3):171-178
doi: 10.3969/j.issn.1672-2531.2006.03.004 |
|
| [5] |
Bian ZX, Li YP, Moher D , et al. Improving the quality of randomized controlled trials in Chinese herbal medicine, Part I: clinical trial design and methodology[J]. Zhong Xi Yi Jie He Xue Bao, 2006,4(2):120-129
doi: 10.3736/jcim20060204 |
|
卞兆祥, 李幼平, David Moher, 等. 提高中草药随机对照试验的质量Ⅰ: 临床试验设计和方法学[J]. 中西医结合学报, 2006,4(2):120-129
doi: 10.3736/jcim20060204 |
|
| [6] | Bian ZX, Moher D, Dagenais S , et al. Improving the quality of randomized controlled trials in Chinese herbal medicine, part IV: applying a revised CONSORT checklist to measure reporting quality[J]. Zhong Xi Yi Jie He Xue Bao, 2006,4(3):233-242 |
| 卞兆祥, David Moher, Simon Dagenais, 等. 提高中草药随机对照试验的质量Ⅳ: 采用修改后的CONSORT条目评价临床随机对照试验报告的质量[J]. 中西医结合学报, 2006,4(3):233-242 | |
| [7] |
Moher D, Schulz KF, Altman DG , et al. The CONSORT statement: revised recommendations for improving the quality of reports of parallel-group randomized trials[J]. JAMA, 2001,285(15):1987-1991
doi: 10.1001/jama.285.15.1987 |
| [8] | Chen HZ. Practice of internal medicine(10th ed)[M]. Beijing: People's Medical Publishing House, 1998,( 10th ed):262 |
| 陈灏珠 . 实用内科学(第10版)[M]. 北京: 人民卫生出版社, 1998,( 第10版):262 | |
| [9] | Chen HZ. Practice of internal medicine(12th ed)[M]. Beijing: People's Medical Publishing House, 2005,( 12th ed):319 |
| 陈灏珠 . 实用内科学(第12版)[M]. 北京: 人民卫生出版社, 2005,( 第12版):319 | |
| [10] | Ministry of Health of the People's Republic of China. Guiding principle of clinical research on new drugs of traditional Chinese medicine[M]. Beijing: Medical Science and Technology Publishing House of China,1997: 71-73 |
| 中华人民共和国卫生部. 中药新药临床研究指导原则[M]. 北京: 中国医药科技出版社,1997: 71-73 | |
| [11] | State Food and Drug Administration . Technology rules of clinical research on new drugs of traditional Chinese medicine[M]. Beijing: Medical Science and Technology Publishing House of China,1999: 2-5 |
| 国家食品药品监督管理局. 中药新药临床研究的技术要求[M]. 北京: 中国医药科技出版社,1999: 2-5 | |
| [12] | Liu YH, Dong JC . Progress in pharmacotherapy of integrated traditional Chinese and Western medicine for virus infection in respiratory tract[J]. Zhong Xi Yi Jie He Xue Bao, 2004,2(3):226-227 |
| 刘羽华, 董竞成 . 呼吸道病毒感染中西医药物治疗进展[J]. 中西医结合学报, 2004,2(3):226-227 | |
| [13] | Pei LX . Research survey of new dosage-form of Chinese herb[J]. Shi Zhen Guo Yi Guo Yao, 2005,16(1):64-65 |
| 裴利霞 . 中药新剂型的研究概况[J]. 时珍国医国药, 2005,16(1):64-65 | |
| [14] |
Zhou JC, Liang YM, Zhou JP . Determination of the andrographolide in andrographolide pill by HPLC[J]. Anhui Yi Yao, 2004,8(4):284-285
doi: 10.3969/j.issn.1009-6469.2004.04.030 |
|
周家才, 梁玉梅, 周久培 . HPLC法测定穿心莲内酯滴丸中穿心莲内酯的含量[J]. 安徽医药, 2004,8(4):284-285
doi: 10.3969/j.issn.1009-6469.2004.04.030 |
|