Journal of Chinese Integrative Medicine ›› 2011, Vol. 9 ›› Issue (2): 201-208.doi: 10.3736/jcim20110214
• Original Experimental Research • Previous Articles Next Articles
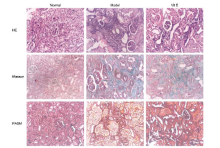
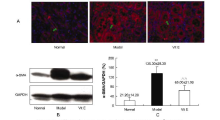
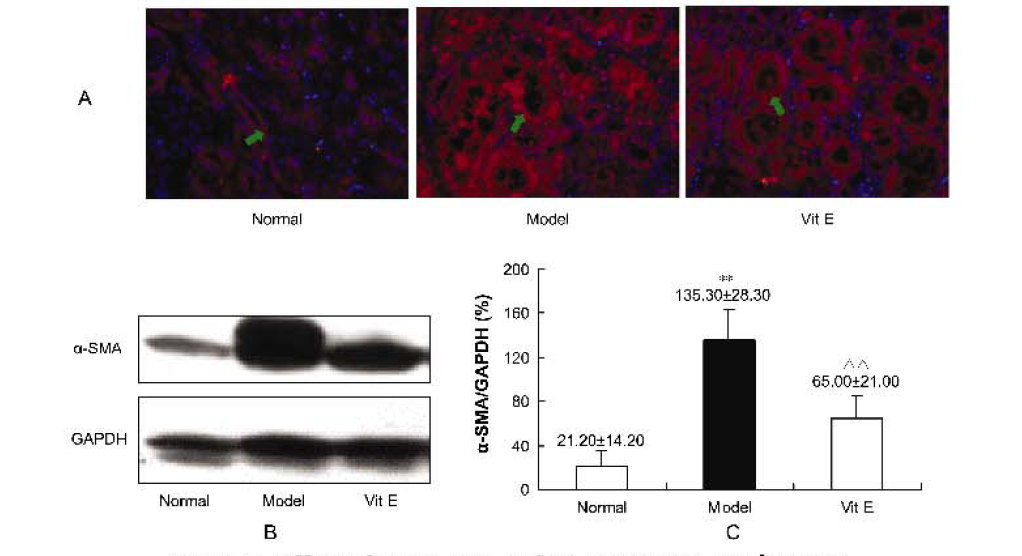
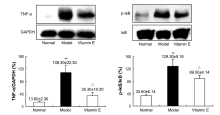
Effects of vitamin E on mercuric chloride-induced renal interstitial fibrosis in rats and the antioxidative mechanism
Yan-yan Tao1, Qing-lan Wang1, Ji-li Yuan1, Li Shen1, Cheng-hai Liu1,2,3,4( )
)
- 1. Institute of Liver Diseases, Shuguang Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
2. Key Laboratory of Liver and Kidney Diseases of Ministry of Education, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
3. E-institute of Internal Medicine of Traditional Chinese Medicine, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
4. Shanghai Key Laboratory of Traditional Chinese Clinical Medicine, Shanghai 201203, China
| [1] |
Ghosh A, Sil PC . A protein from Cajanus indicus Spreng protects liver and kidney against mercuric chloride-induced oxidative stress[J]. Biol Pharm Bull, 2008,31(9):1651-1658
doi: 10.1248/bpb.31.1651 |
| [2] |
Vahter M, Akesson A, Lind B , Björs U , Schütz A , Berglund M.Longitudinal study of methylmercury and inorganic mercury in blood and urine of pregnant and lactating women, as well as in umbilical cord blood[J]. Environ Res, 2000,84(2):186-194
doi: 10.1006/enrs.2000.4098 |
| [3] |
Richet G, Ducrot H, Hamburger J, Legrain M, Maxwell M, Péquignot MH . May medical therapy of arterial hypertension by used in patients with renal failure[J]. Presse Med., 1968,76(44):2103-2106
doi: 10.1155/2015/208519 pmid: 5701039 |
| [4] |
Liu Y . Epithelial to mesenchymal transition in renal fibrogenesis: pathologic significance, molecular mechanism, and therapeutic intervention[J]. J Am Soc Nephrol, 2004,15(1):1-12
doi: 10.1097/01.ASN.0000106015.29070.E7 |
| [5] |
Nielsen JB, Andersen O . Oral mercuric chloride exposure in mice: effects of dose on intestinal absorption and relative organ distribution[J]. Toxicology, 1989,59(1):1-10
doi: 10.1016/0300-483X(89)90152-2 |
| [6] |
Nielsen JB, Andersen O . Disposition and retention of mercuric chloride in mice after oral and parenteral administration[J]. J Toxicol Environ Health, 1990,30(3):167-180
doi: 10.1080/15287399009531420 pmid: 2366256 |
| [7] | Yuan JL, Liu CH, Jiang ZH, Zhang Y, Liu C . Effects of oral administration of HgCl2 on renal interstitial fibrosis in rats[J]. Zhongguo Shi Yan Dong Wu Xue Bao, 2006,14(2):93-95 |
| 袁继丽, 刘成海, 姜哲浩, 张悦, 刘成 . 口服氯化汞对大鼠肾间质纤维化的作用[J]. 中国实验动物学报, 2006,14(2):93-95 | |
| [8] |
Yuan JL, Liu CH, Zhang Y, Jiang ZH, Liu C . The mechanism of lipid peroxidation in renal interstitial fibrosis in rats intoxicated by mercuric chloride[J]. Zhongguo Zhong Xi Yi Jie He Shen Bing Za Zhi, 2008,9(5):403-407
doi: 10.3969/j.issn.1009-587X.2008.05.008 |
|
袁继丽, 刘成海, 张悦, 姜哲浩, 刘成 . 氯化汞诱导大鼠肾间质纤维化的脂质过氧化损伤机制研究[J]. 中国中西医结合肾病杂志, 2008,9(5):403-407
doi: 10.3969/j.issn.1009-587X.2008.05.008 |
|
| [9] |
Coates ME, Kon SK, Porter JW . Vitamins in animal nutrition[J]. Br Med Bull, 1956,12(1):61-66
doi: 10.1093/oxfordjournals.bmb.a069518 |
| [10] |
Bennett RT, Mazzaccaro RJ, Chopra N, Melman A, Franco I . Suppression of renal inflammation with vitamins A and E in ascending pyelonephritis in rats[J]. J Urol, 1999,161(5):1681-1684
doi: 10.1097/00005392-199905000-00089 pmid: 10210439 |
| [11] | Qiu CJ, Wang HF, Wei D . Effect of vitamin E on advanced glycosylation end-products and their receptors in renal tissue of diabetic rats[J]. Xi’an Jiao Tong Da Xue Xue Bao Yi Xue Ban, 2008,29(3):297-300 |
| 邱春娟, 王惠芳, 魏动 . 维生素E对糖尿病大鼠高级糖基化终末产物及其受体和肾功的影响[J]. 西安交通大学学报(医学版), 2008,29(3):297-300 | |
| [12] |
Jamall IS, Finelli VN, Que Hee SS . A simple method to determine nanogram levels of 4-hydroxyproline in biological tissues[J]. Anal Biochem, 1981,112(1):70-75
doi: 10.1016/0003-2697(81)90261-X pmid: 7258630 |
| [13] |
Fukagawa M, Noda M, Shimizu T, Kurokawa K . Chronic progressive interstitial fibrosis in renal disease — are there novel pharmacological approaches[J]. Nephrol Dial Transplant, 1999,14(12):2793-2795
doi: 10.1093/ndt/14.12.2793 |
| [14] |
Eddy AA . Molecular basis of renal fibrosis[J]. Pediatr Nephrol, 2000,15(3-4):290-301
doi: 10.1007/s004670000461 pmid: 11149129 |
| [15] |
Liu Y . Renal fibrosis: new insights into the pathogenesis and therapeutics[J]. Kidney Int, 2006,69(2):213-217
doi: 10.1038/sj.ki.5000054 pmid: 16408108 |
| [16] | Nielsen JB, Andersen HR, Andersen O, Starklint H . Dose and time relations in Hg++-induced tubular necrosis and regeneration[J]. Environ Health Perspect, 1994,102(Suppl 3):317-320 |
| [17] |
Guillermina G, Adriana TM, Monica EM . The implication of renal glutathione levels in mercuric chloride nephrotoxicity[J]. Toxicology, 1989,58(2):187-195
doi: 10.1016/0300-483X(89)90008-5 |
| [18] |
Berndt WO, Baggett JM, Blacker A, Houser M . Renal glutathione and mercury uptake by kidney[J]. Fundam Appl Toxicol, 1985,5(5):832-839
doi: 10.1016/0272-0590(85)90166-6 pmid: 4065459 |
| [19] |
Yonaha M, Ohbayashi Y, Ichinose T, Sagai M . Lipid peroxidation stimulated by mercuric chloride and its relations to the toxicity[J]. Chem Pharm Bull (Tokyo), 1982,30(4):1437-1442
doi: 10.1248/cpb.30.1437 pmid: 6213317 |
| [20] |
Fukino H, Hirai M, Hsueh YM, Yamane Y . Effect of zinc pretreatment on mercuric chloride-induced lipid peroxidation in the rat kidney[J]. Toxicol Appl Pharmacol, 1984,73(3):395-401
doi: 10.1016/0041-008X(84)90091-7 pmid: 6232736 |
| [21] |
Elks CM, Mariappan N, Haque M, Guggilam A, Majid DS, Francis J . Chronic NF-κB blockade reduces cytosolic and mitochondrial oxidative stress and attenuates renal injury and hypertension in SHR[J]. Am J Physiol Renal Physiol, 2009,296(2):F298-F305
doi: 10.1152/ajprenal.90628.2008 |
| [22] |
Elsharkawy AM, Mann DA . Nuclear factor-κB and the hepatic inflammation-fibrosis-cancer axis[J]. Hepatology, 2007,46(2):590-597
doi: 10.1002/(ISSN)1527-3350 |
| [23] |
Wu ZH, Miyamoto S . Many faces of NF-κB signaling induced by genotoxic stress[J]. J Mol Med, 2007,85(11):1187-1202
doi: 10.1007/s00109-007-0227-9 |
| [24] |
Karin M, Ben-Neriah Y . Phosphorylation meets ubiquitination: the control of NF-κB activity[J]. Annu Rev Immunol, 2000,18:621-663
doi: 10.1146/annurev.immunol.18.1.621 pmid: 10837071 |
| [25] |
Adcock IM, Brown CR, Kwon O, Barnes PJ . Oxidative stress induces NFκB DNA binding and inducible NOS mRNA in human epithelial cells[J]. Biochem Biophys Res Commun, 1994,199(3):1518-1524
doi: 10.1006/bbrc.1994.1403 |
| [26] |
Cheng S, Lovett DH.Gelatinase A ( MMP-2) is necessary and sufficient for renal tubular cell epithelial-mesenchymal transformation[J]. Am J Pathol, 2003,162(6):1937-1949
doi: 10.1016/S0002-9440(10)64327-1 |
|